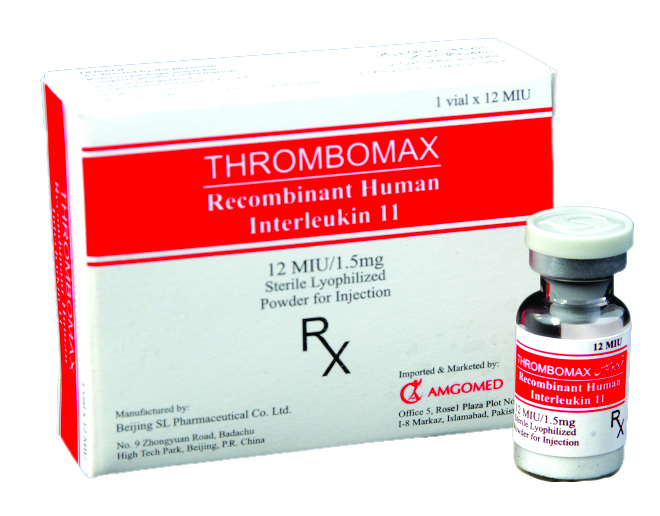
Thrombomax (IL-11) is a new treatment option which is FDA approved & now available in Pakistan for Thrombocytopenic patients.
Thrombomax, Interleukin eleven (IL-11) is a thrombopoietic growth factor that directly stimulates the proliferation of hematopoietic stem cells and megakaryocyte progenitor cells and induces megakaryocyte maturation resulting in increased platelet production. IL-11 is a member of a family of human growth factors. Oprelvekin, the active ingredient in Thrombomax, is produced in Escherichia coli (E. coli) by recombinant DNA technology. Trials have shown that mature megakaryocytes which develop during in vivo treatment with Thrombomax are ultra-structurally normal and possessed a normal life span.
Thrombomax is indicated for the prevention of severe thrombocytopenia and the reduction of the need for platelet transfusions following myelosuppressive chemotherapy in adult patients with non-myeloid malignancies who are at high risk of severe thrombocytopenia. Efficacy was demonstrated in patients who had experienced severe thrombocytopenia following the previous chemotherapy cycle. Thrombomax is not indicated following myeloablative chemotherapy.
Mean time to platelet recovery to ≥20,000 cells/µL with rhIL-11 is at least 2.5 days vs 6.4 days for placebo patients. [3]
Mean time to platelet recovery to ≥ 50,000 cells/µL with rhIL-11 is at least 9.3 days vs 13.0 days with placebo. [3]
Thrombocytopenia may compromise cancer treatment, causing chemotherapy dose reductions, schedule alterations, or the need for platelet transfusions.
Read more
Colorectal cancer is a major cause of morbidity and mortality, being one of the most common malignant tumors in the world.
Read more
Cancer of the prostate is the most common malignancy in elderly men
Read more
Chemotherapy-induced neutropenia is a common and serious complication of myelosuppressive chemotherapy. It is associated with significant morbidity and mortality, and can increase the overall cost of providing cancer therapy.
Read more
Some types of cancer may cause bone damage leading to high levels of blood calcium and lesions that cause severe bone pain, bone masses & fractures.
Read more